AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Samtools get consensus sequences9/14/2023  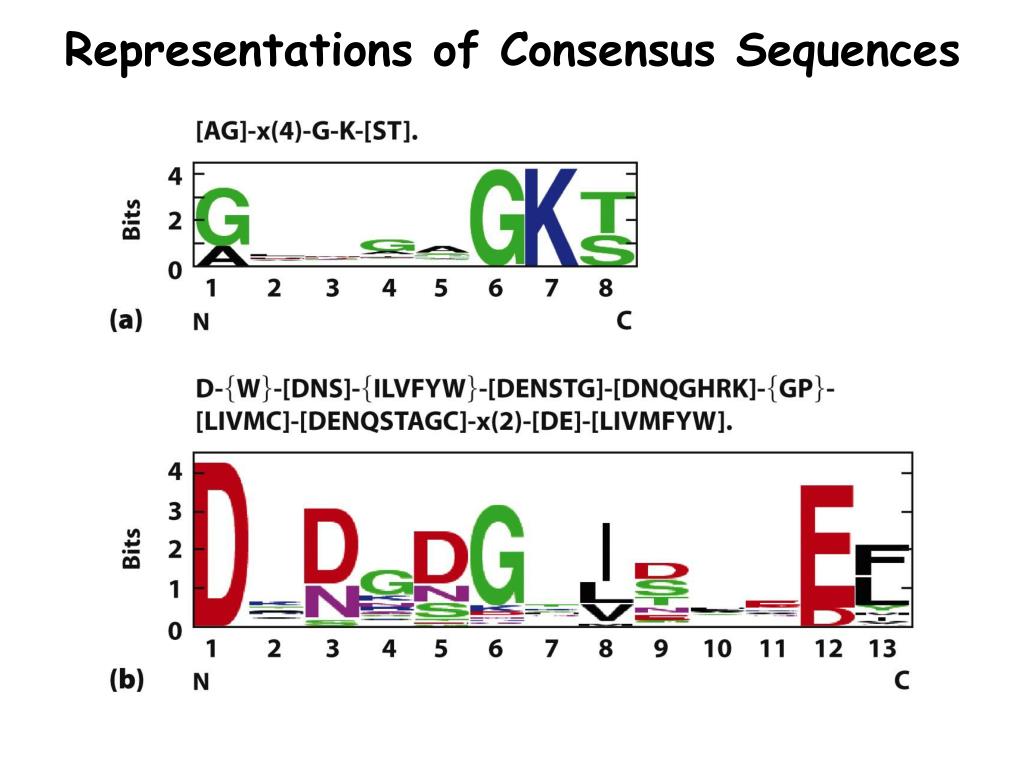
Take a look here for a detailed manual page for each function in samtools. Sorted, indexed BAM files are filteredbased on location, flags, mapping quality ( samtools view with filtering options).Sorted BAM files are indexed ( samtools index).BAM files are sorted by reference coordinates ( samtools sort).SAM files are converted into BAM files ( samtools view).Most functionality while using BAM files can be described as such: Since most aligners produce a BAM file, we'll work on some basic manipulations of the BAM files we produced from our alignments yesterday. We'll be focusing on just a few of samtoolsfunctions in this series of exercises. F 0xXX only report alignment records where the. f 0xXX only report alignment records where the specified flags are all set (are all 1) you can provide the flags in decimal, or as here as hexadecimal. This is a good way to remove low quality reads, or make a BAM file restricted to a single chromosome. The most common samtools view filtering options are: -q N only report alignment records with mapping quality of at least N ( > N ). bam files - they can be converted into a non-binary format ( SAM format specification here) and can also be ordered and sorted based on the quality of the alignment. These files are compressed, so they can't be viewed using standard unix file viewers such as more, less and head. The typical segment length is determined by finding the median length of the segment/subject reference sequences whose contig alignments have the highest bitscore.Introduction to Samtools - manipulating and filtering bam filesĪs we showed you yesterday, the main type of output from aligning reads to a databases is a binary alignment file, or BAM file. Segment_cov : the number of sequenced bases in the consensus sequence divided by the typical length of this genome segment (as a percentage). Sequenced_bases : the number of nucleotide positions in the consensus sequence with sufficient depth of coverage (set by -D argument) and a succesful base call (e.g. Seq_length : the length (in nucleotides) of the consensus sequence generated by FluViewer 
Mapped reads : the number of sequencing reads mapped to this segment Subtype : HA or NA subtype ("none" for internal segments) Segment : influenza A virus genome segment (PB2, PB1, PA, HA, NP, NA, M, NS) The report TSV file contains the following columns:Ĭonsensus_seq : the name of the consensus sequence described by this row Headers in the FASTA file have the following format: >output_name_unique_sequence_number|segment|subject A report TSV file describing segment, subtype, and sequencing metrics for each consensus sequence.A sorted BAM file with reads mapped to either the choosen reference sequences (align mode) or the assembled contigs (assembly mode).How to count the number of mapped reads in a BAM or SAM file (SAM bitcode fields) more statistics about alignments. A FASTA file containing consensus sequences for influenza A virus genome segments get number of individual reads, paired reads that mapped both count double R1+R2.Headers for these sequences must be formatted and annotated as follows: >unique_id|strain_name|segment|subtypeįor example: >MF599463|A/swine/Kansas/A01378028/2017|HA|H3 g : Set this flag to deactivate garbage collection and retain intermediate files FluViewer DatabaseįluViewer requires a curated FASTA file "database" of influenza A virus reference sequences. i : Minimum nucleotide sequence identity between database reference sequence and contig (percentage, default = 95) c : Minimum coverage of database reference sequence by contig (percentage, default = 25) q : Minimum PHRED score for base quality and mapping quality (default = 30) D : Minimum read depth for base calling (default = 20) m : FluViewer run mode (align or assemble) 
o : output name (creates directory with this name for output, includes this name in output files, and in consensus sequence headers) d : path to FASTA file containing FluViewer database (details below) r : path to FASTQ file containing reverse reads f : path to FASTQ file containing forward reads Custom DBs can be created and used as well (instructions below).
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
